When comparing Ozempic and Mounjaro, the primary distinction lies in their mechanism of action and resulting effectiveness for weight management. Both are MHRA-approved, prescription-only injections, but clinical trial data indicates that Mounjaro’s dual-action mechanism often leads to more significant weight loss compared to Ozempic’s single-hormone approach. Ultimately, the most suitable choice for a patient is determined by a thorough clinical assessment of their individual health profile and treatment goals.
A Clinical Overview of Ozempic and Mounjaro
Understanding the available weight management treatments can feel complex. Ozempic (semaglutide) and Mounjaro (tirzepatide) are two effective medications available in the UK for managing both type 2 diabetes and supporting weight management. Both are administered as a once-weekly injection and work by mimicking gut hormones that help to regulate appetite and blood glucose levels.
However, they are not interchangeable. The crucial difference is in their active ingredients and how they interact with the body's hormone receptors.
- Ozempic (semaglutide) is a GLP-1 receptor agonist. It targets a single, powerful pathway involved in appetite regulation.
- Mounjaro (tirzepatide) is a dual GLP-1 and GIP receptor agonist. This means it targets two different pathways, which can produce a more comprehensive effect on metabolism and hunger signals.
This fundamental pharmacological difference explains the varying results observed in clinical studies. It is also why both are strictly prescription-only medications. A UK-registered prescriber must conduct a full clinical review to determine if either treatment is safe and suitable for an individual. Sourcing these treatments through a regulated provider, such as a UK-registered pharmacy, is essential for ensuring patient safety and adherence to healthcare protocols.
Ozempic vs Mounjaro At a Glance
This table provides a basic comparison of these two prescription-only treatments.
| Feature | Ozempic (Semaglutide) | Mounjaro (Tirzepatide) |
|---|---|---|
| Active Ingredient | Semaglutide | Tirzepatide |
| Mechanism | Mimics one hormone (GLP-1) | Mimics two hormones (GLP-1 & GIP) |
| Licensed Use | Type 2 Diabetes (used off-label for weight) | Type 2 Diabetes & Weight Management |
| Administration | Once-weekly self-injection | Once-weekly self-injection |
| Regulatory Status | MHRA-approved | MHRA-approved |
While they share similarities, such as the weekly injection schedule, Mounjaro's dual-hormone action is the key differentiator that often translates to different clinical outcomes.
How Ozempic and Mounjaro Work Differently
To understand the core of the Ozempic vs Mounjaro comparison, it is necessary to examine their biological mechanisms. While both are injectable, prescription-only medicines that help manage appetite and blood glucose, their mechanisms are distinct. This difference is why varying results are seen in clinical trials.
Both medications are classified as incretin mimetics. In simple terms, they mimic the natural hormones released by the gut after a meal. These hormones are vital messengers, influencing insulin release, digestion speed, and signals of fullness sent to the brain.
The Single-Action Approach of Ozempic
Ozempic’s active ingredient is semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist. This means it selectively activates the receptors for one specific hormone: GLP-1.
When you eat, the gut naturally releases GLP-1, which initiates several important processes:
- It stimulates insulin release, helping the body to use glucose from food and prevent blood sugar spikes.
- It slows gastric emptying, which contributes to a feeling of fullness for a longer duration.
- It sends satiety signals to the hypothalamus—the brain's appetite control centre—thereby reducing appetite.
By imitating this single hormone, Ozempic enhances these natural bodily functions, which can lead to improved blood glucose control and reduced calorie intake.
Mounjaro and Its Dual-Hormone Mechanism
Mounjaro employs a different mechanism. Its active ingredient, tirzepatide, is a first-in-class dual-agonist, meaning it targets two different types of gut hormone receptors.
Mounjaro acts as both a GLP-1 receptor agonist (like Ozempic) and a glucose-dependent insulinotropic polypeptide (GIP) receptor agonist. This dual action is the key differentiator and is understood to be the reason for its efficacy in weight management trials.
GIP is another important incretin hormone. It also stimulates insulin release but has a distinct, complementary role in energy balance and fat metabolism. By activating both GLP-1 and GIP pathways simultaneously, Mounjaro creates a powerful, synergistic effect.
Instead of replicating the effects of a single hormone, it amplifies the body’s entire post-meal response by engaging two primary components of the metabolic system. This combined action on appetite and metabolism appears to lead to more profound appetite suppression and greater improvements in how the body processes sugar and fat. This distinction is vital when discussing Ozempic vs Mounjaro, as it underpins why one may be more clinically effective for certain patients. For further details on this prescribed medication, you can learn more about Mounjaro and its specific uses.
The choice between a single- or dual-agonist medication is a clinical one. This decision is made by a qualified prescriber following a thorough assessment, which any UK-registered pharmacy will ensure is completed before supplying a prescription-only treatment.
A Review of Clinical Trial Data on Weight Loss
The most robust evidence in the Ozempic versus Mounjaro comparison comes directly from clinical trials. These large-scale studies provide a clear, data-driven picture of what each medication can achieve for weight loss. While both are effective, the data consistently show that Mounjaro’s dual-action mechanism gives it an advantage, often leading to more significant weight reduction.
This section reviews the key clinical trial data. It is important to remember that these are prescription-only medicines, obtainable only through a regulated provider after a full clinical assessment.

Key Findings from the SURPASS and SURMOUNT Trials
The evidence supporting Mounjaro (tirzepatide) is based on two major sets of trials: the SURPASS programme for type 2 diabetes and the SURMOUNT programme for weight management.
The SURPASS-2 trial was particularly informative as it was a direct, head-to-head comparison of tirzepatide against semaglutide (the active drug in Ozempic). In patients with type 2 diabetes, those on the highest dose of tirzepatide experienced greater reductions in both blood glucose and body weight than those on the highest dose of semaglutide.
For individuals without diabetes, the SURMOUNT trials highlighted the drug's weight loss efficacy.
- SURMOUNT-1: This was a landmark study involving adults living with obesity or who were overweight with at least one related health condition.
- The Results: After 72 weeks, participants on the highest dose of Mounjaro (15 mg) lost an average of 22.5% of their initial body weight.
This represented a significant advancement in pharmacological weight management.
How Does Semaglutide’s Performance Compare?
Semaglutide, the drug in Ozempic, also has a strong evidence base from its own series of trials, known as the STEP programme. Although Ozempic is licensed for type 2 diabetes, these trials investigated semaglutide purely for weight management, which led to the approval of Wegovy (a higher-dose version of semaglutide).
In the STEP 1 trial, participants without diabetes taking a 2.4 mg weekly dose of semaglutide—the dose used in Wegovy—lost an average of 14.9% of their body weight over 68 weeks. It is important to note that the highest Ozempic dose licensed for diabetes in the UK is 1.0 mg, which generally results in less weight loss than the higher Wegovy dose. You can explore this further in our guide to Wegovy and its clinical use.
In summary: In both head-to-head studies and separate major trials, Mounjaro (tirzepatide) has consistently demonstrated a higher average percentage of total body weight loss compared to semaglutide, particularly at the maximum approved doses.
Understanding the Dose-Dependent Effect
An important concept when comparing Ozempic and Mounjaro is that their effects are dose-dependent. For both drugs, higher doses tend to produce greater weight loss. The key difference is that Mounjaro's potential for weight loss appears to have a higher ceiling.
In the SURMOUNT-1 study, even participants on the lowest dose of Mounjaro (5 mg) lost an average of 16% of their body weight. This figure is slightly greater than the average loss observed with the highest dose of semaglutide in its key trial.
This suggests that Mounjaro’s dual-agonist mechanism, targeting both GLP-1 and GIP receptors, provides a more potent effect for weight reduction across its dosing schedule. A clinician at a UK-registered pharmacy will carefully consider this data, alongside a patient's personal health profile, to determine the appropriate starting dose and titration plan.
Ultimately, while both are highly effective, MHRA-approved treatments, the clinical evidence indicates that Mounjaro holds a statistically significant advantage for achieving maximum weight loss. This is why a thorough consultation with a healthcare professional is vital—it ensures the treatment’s potential is matched to individual health goals safely.
Understanding Side Effects and Safety Profiles
When considering a new medicine, understanding its safety profile is essential. Both Ozempic and Mounjaro have been approved for use in the UK by the MHRA, meaning they are considered safe and effective when prescribed and managed appropriately by a healthcare professional. However, like any effective medication, they are associated with potential side effects.
Patient safety is the highest priority. Before any prescription is issued, a thorough clinical assessment is required to ensure the treatment is suitable for an individual's specific health circumstances. This process identifies any potential risks or contraindications.

Common Gastrointestinal Side Effects
The most common side effects for both Ozempic and Mounjaro are gastrointestinal. This is expected, given their mechanism of action involves slowing gastric emptying.
These side effects are typically most prominent at the start of treatment or following a dose increase. For most people, symptoms like nausea or diarrhoea are mild to moderate and tend to resolve within a few weeks as their body adapts.
The most frequently reported side effects include:
- Nausea: A feeling of sickness is the most common side effect for both drugs.
- Diarrhoea: Changes in bowel habits are also common.
- Constipation: As digestion slows, some individuals may experience constipation.
- Vomiting: This is less common than nausea but can occur, particularly at higher doses.
- Abdominal Discomfort: Symptoms may include bloating, flatulence, or general stomach pain.
A clinician will manage these effects by starting treatment at the lowest dose and increasing it gradually. Simple measures, such as maintaining adequate hydration and consuming smaller, more frequent meals, can also help.
Comparing Side Effect Incidence
While the types of side effects are very similar, clinical trial data show slight differences in their frequency. Mounjaro's dual-action mechanism, which contributes to its high efficacy, may also lead to a slightly higher incidence of these gastrointestinal issues, particularly at its higher doses.
A key finding from the studies is that although Mounjaro may be associated with a slightly higher rate of gut-related side effects, the vast majority are mild. Importantly, very few participants in the major trials discontinued either medication due to side effects, indicating they are generally well-tolerated.
The table below provides a general overview based on large clinical trials. Individual experiences can vary.
Common Side Effect Comparison
This table offers a general comparison of how frequently common side effects were reported in clinical trials for each medication.
| Side Effect | Ozempic (Semaglutide) Incidence | Mounjaro (Tirzepatide) Incidence |
|---|---|---|
| Nausea | Reported by 15-20% of users, dose-dependent. | Reported by 20-30% of users, particularly at higher doses. |
| Diarrhoea | Occurs in approximately 8-9% of patients. | Occurs in approximately 12-17% of patients. |
| Constipation | Affects around 5-7% of users. | Affects around 6-11% of users. |
| Vomiting | Reported by 5-9% of participants. | Reported by 8-13% of participants. |
As shown, the incidence rates for Mounjaro are consistently slightly higher, which a clinician will consider when making a recommendation.
Serious but Rare Risks
Beyond the common side effects, there are several more serious but much rarer risks. It is vital to discuss these with a prescriber so they can assess personal risk factors during the consultation.
These include:
- Pancreatitis: Inflammation of the pancreas. If severe, persistent abdominal pain occurs, the medication should be stopped and immediate medical attention sought.
- Gallbladder Problems: Issues such as gallstones or cholecystitis have been reported.
- Thyroid C-cell Tumours: Both drugs carry a warning about a potential risk of thyroid C-cell tumours. This is based on studies in rodents, and it is not known if this risk applies to humans. Due to this theoretical risk, neither medication should be used by anyone with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
This underscores the importance of providing a complete and accurate medical history. Reputable providers, regulated by the GPhC, conduct these stringent checks before prescribing. The clinician will then provide ongoing support to help manage any side effects that may arise.
How to Access Treatment in the UK
In the UK, there are two primary pathways to obtain a prescription for Ozempic or Mounjaro: through the National Health Service (NHS) or via a private healthcare provider. Both pathways are strictly regulated to ensure these medications are used safely and appropriately, but the eligibility criteria differ significantly.
Both Ozempic and Mounjaro are fully approved by the MHRA, the UK's medicines regulator, having met rigorous standards for safety, quality, and effectiveness. However, this approval does not permit open access. A full consultation with a UK-registered prescriber is always required to assess a patient's health and determine if a prescription is appropriate.
NHS Eligibility Criteria
Access to these treatments on the NHS is governed by guidelines from the National Institute for Health and Care Excellence (NICE). The criteria are highly specific, targeting patients with the greatest clinical need, typically those with type 2 diabetes or severe weight-related health complications.
For weight management, eligibility generally requires:
- A very high Body Mass Index (BMI), typically 35 kg/m² or more.
- At least one serious health condition linked to weight, such as pre-diabetes or cardiovascular disease.
- Care under a specialist weight management service.
Due to these strict criteria, many individuals who could potentially benefit from these medications may not qualify for NHS treatment.
The Private Healthcare Pathway
For individuals who meet the clinical criteria for treatment but do not meet the narrow NHS guidelines, the private route is the main alternative. This includes services from a regulated online pharmacy, which provides a safe and direct way to receive a clinical assessment and, if appropriate, a prescribed medication.
This pathway allows individuals with a qualifying BMI (often lower than the NHS threshold) to access treatment following a health assessment with a clinician. All legitimate private providers, including any UK-registered pharmacy, are regulated by the General Pharmaceutical Council (GPhC), ensuring adherence to the same high standards of patient safety and care.
Navigating Supply and Choosing a Provider
The global demand for Ozempic and Mounjaro has led to intermittent supply shortages, which can affect availability through both NHS and private channels. When choosing a provider, it is crucial to select one that is transparent about stock levels and is fully regulated. A reputable provider will always prioritise a comprehensive clinical consultation over simply issuing a prescription.
By working with a regulated service, patients are assured of:
- A thorough assessment by a UK-registered prescriber.
- Genuine, MHRA-approved medication.
- Continuous clinical support and monitoring.
- Clear guidance on dosing, managing side effects, and implementing lifestyle changes.
This structured, professional approach ensures treatment is not only effective but, most importantly, safe.
Which Treatment Is Right for You?
The decision between Ozempic and Mounjaro is a clinical one, made in collaboration with a qualified UK prescriber following a detailed review of your health, medical history, and treatment objectives. The "best" choice is always the one that is safest and most clinically appropriate for the individual.
A clinician will carefully evaluate several key factors. While both are effective, MHRA-approved treatments, their distinct profiles may make one a better fit for a patient's specific circumstances. The goal is to align the medication's characteristics with unique health needs.
Prioritising Efficacy and Clinical Need
For individuals with a greater amount of weight to lose or with specific metabolic health concerns, a clinician might consider Mounjaro as a primary option. Its dual-agonist mechanism, acting on both GLP-1 and GIP receptors, has consistently produced more substantial weight loss results in head-to-head clinical trials.
If achieving the greatest possible weight reduction safely is the primary objective, the compelling data from the SURMOUNT trials often places Mounjaro at the forefront of the clinical discussion. A prescriber will assess if this higher level of efficacy is a priority for managing an individual's weight-related health conditions.
This does not diminish the value of Ozempic, which is a highly effective medication with a long-standing safety record. Past experiences with similar treatments, or a clinical preference for initiating treatment with a single-agonist medication, could make Ozempic the appropriate initial choice.
Considering Side Effect Tolerance and Medical History
An individual's potential tolerance for side effects is another crucial consideration. The side effect profiles of Ozempic and Mounjaro are very similar, primarily involving gastrointestinal issues like nausea. However, some evidence suggests these symptoms may be slightly more common with Mounjaro, particularly at higher doses.
A clinician will discuss this with the patient. If an individual has a history of sensitivity to medications or is particularly concerned about side effects, the treatment plan may prioritise tolerability. This could influence not only the choice of drug but also the rate at which the dose is increased.
A complete medical history is non-negotiable. Certain conditions, such as a personal or family history of specific thyroid cancers or pancreatitis, are contraindications for both medications. This is why a detailed, honest consultation through a regulated service, such as a UK-registered pharmacy, is essential for patient safety.
This flowchart illustrates the two main pathways for accessing these treatments in the UK: via the NHS or a private provider.
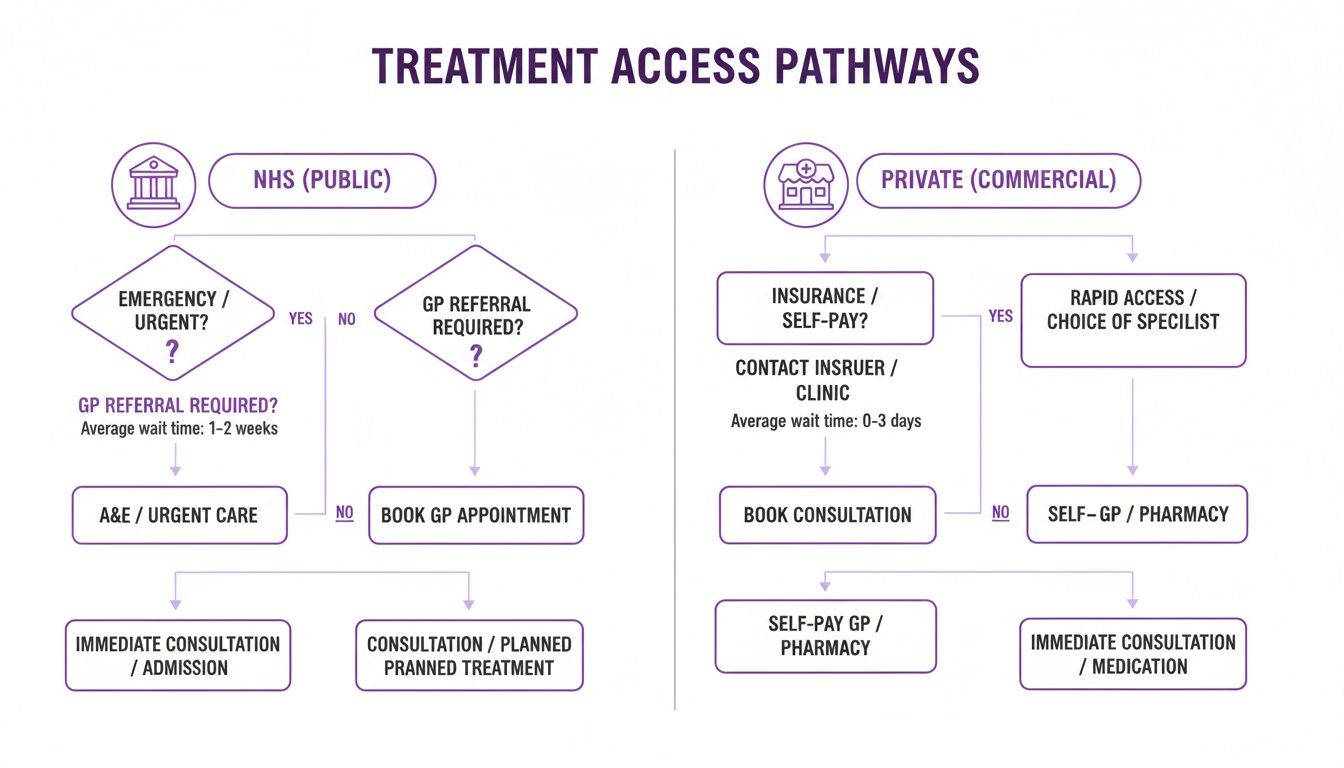
As the graphic shows, both pathways are regulated, but the eligibility criteria and timelines can vary considerably.
Ultimately, the choice between Ozempic and Mounjaro is based on a careful balance of efficacy, safety, and personal circumstances. This personalised approach, guided by a healthcare professional from a regulated online pharmacy, ensures the prescription-only treatment received is optimised for an individual's health journey.
Frequently Asked Questions
It is natural to have questions when considering a new treatment like Ozempic or Mounjaro. Here are some straightforward, clinically informed answers to common queries. This information should be seen as a starting point for a more detailed discussion with a clinician.
How often do I need to inject Ozempic and Mounjaro?
Both medications are administered as a once-weekly injection under the skin (subcutaneously).
Common injection sites include the abdomen, thigh, or the back of the upper arm. The weekly schedule helps to maintain a consistent level of the medication in the body. A clinician will provide full instructions on the correct injection technique before treatment begins.
What should I do if I miss a dose?
If a dose is missed, the guidance is as follows: if it has been 5 days or less since the scheduled dose, inject the missed dose as soon as you remember. Then, take the next dose on the usual scheduled day.
If it has been more than 5 days, skip the missed dose entirely. Do not take two doses at once. Wait and take the next dose on its regularly scheduled day.
Is it possible to switch from Ozempic to Mounjaro, or vice versa?
Yes, switching between these medications is possible, but it must be managed by a prescriber. It should never be done without medical supervision.
A clinician will evaluate treatment progress, tolerability, and overall health goals to determine if a switch is appropriate. They will then create a safe transition plan, which may involve a washout period or starting the new medication at its lowest dose.
Do these injections replace diet and exercise?
No. These medications are most effective when used as part of a comprehensive management plan that includes a balanced diet and regular physical activity.
Ozempic and Mounjaro can help to manage appetite and cravings, making adherence to a calorie-controlled diet more achievable. For sustainable results, they should be combined with long-term lifestyle modifications.
Will the weight return if I stop the medication?
This is an important consideration. As these treatments actively manage appetite and metabolism, discontinuing them can lead to a return of previous hunger cues and, potentially, weight regain.
The key to maintaining weight loss is to establish sustainable healthy habits during treatment. A prescriber from a GPhC-regulated online pharmacy will work with you to develop a long-term plan, helping you to maintain your progress after the medication is stopped.



0 comments